Lung Consolidation and Cardio Pulmonary Rehabilitation
- yiannis1013
- Nov 12, 2024
- 36 min read
Lung Consolidation and Cardio Pulmonary Rehabilitation
The Case Study and Mechanical Hypothesis
An interesting case of exercise rehabilitation for lung consolidation secondary to infection, known as Acute Respiratory Distress Syndrome (ARDS). Subject requires a return to manual work to include rehabilitation of cardiovascular
capacity and some functional Power.
ARDS is a serious lung condition that causes low blood oxygen and results in widespread
pulmonary edema formation. More precisely it occurs when fluid builds up in the small air sacs (alveoli) of the lungs (lung consolidation), and the alveoli epithelial layer breaks down. Mechanisms of lung injury that lead to loss of the epithelium and alveolar fluid barrier function centre on exuberant non-resolving inflammation, barrier dysfunction, and surfactant system
impairement (Zuttion et al., 2022; Sanches et al., 2022).
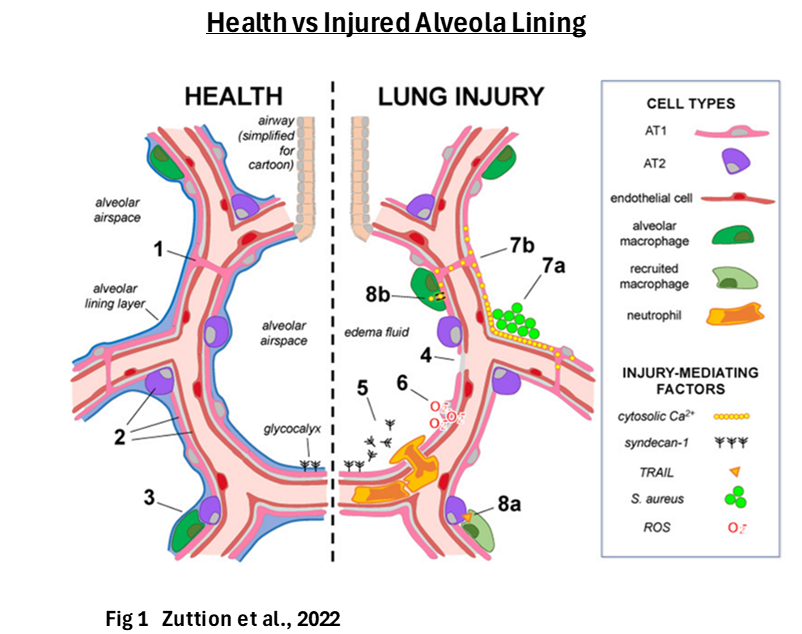
Damage to the epithelial lining and Permeability
The hypothesised issue at hand is how to increase lung capacity and functional fitness, while optimising for fluid clearance from the lungs. This concern is derived from the mechanical characteristics of fluid regulation in the lungs, primarily the alveoli. There are two primary mechanisms of fluid regulation; osmosis and active transport:
Osmosis
There is natural variability in the amount of fluid that may defuse into the alveola as governed by Starling’s forces (Miserocchi and Beretta, 2022).
Starling’s forces explain the dynamics of hydrostatic pressure and colloid osmotic pressure (oncotic pressure); i.e. passive diffusion (O’Brodovich, 2001). This rate of diffusion is also effected by the structural integrity of the epithelial barrier and other physiological states (e.g. exercise intensity).
Active Transport
Fluid Removal from the alveoli is a function of active (Na+) transport and a separable mechanism to the ‘Starling’s forces’ associated diffusion capacity. This too is affected by the structural and other physiological states (e.g. inflammation).
The severity of lung edema can be described as being dependent on the competing increases in permeability, and the active clearance of edema from the air spaces (Frank and Matthey, 2014); Tedjasaputra, 2016). Acute or chronic lung injury, exercise stress and other physiological events, are all modulatory factors in the balance of this system of passive diffusion and active clearance.
Edema, Diffusion Capacity, Adaption and Exercise
Edema
Although healthy lung function has been observed to be resistant to edema (Effros and Parker, 2009), aerobic exercise even in the non-injured lung is inherently edemagenic due to the increase in cardiac output and the resultant increase in haemodynamic pressure (Miserocchi and Beretta, 2022). During acute exercise bouts, resultant increases in blood pressure and haemodynamic blood flow will increase arterial pressure and thus the the rate of defusion due to these changes in osmotic pressure. This is the direct result of high pulmonary capillary hydrostatic pressure that induces fluid movement into the interstitial and alveolar spaces (Fermoyle et al., 2020). It can therefore be inferred that aerobic exercise has the capacity to increase lung (alveola) edema, and more specifically in intense exercise as has been shown in case and control studies, see Bates et al., (2011) as one example.
Miserocchi and Beretta (2023) have discussed observed correlations (Zavorsky, 2007) based on intensity of exercise more broadly. Submaximal exercise as well as the incremental V̇O2 max tests not exceeding 20–25 min time, were found not sufcient to trigger pulmonary edema. In the case of prolonged exercise at 50–75% V̇O2 max, and ranging from 15 min up to 2 h, 16% of subjects were discribed as potentially showing signs of edema (Miserocchi and Beretta, 2023; Zavorsky, 2007). A cautionary interpretation on the low intensity endurance data should be applied (also see fig 2):
1. Manier et al., (1999) did not measure edema, and found no significant alteration in pulmonary density after the 2-h run test. Lung volume slightly increased, and lung mass remained stable because of a change in density distribution.
2. Hodges et al., (2005) performed 1 h of exercise at 77% (near lactate threshold) of maximal O2. Higher concentrations of surfactant apoprotein A and a higher surfactant apoprotein A-to-phospholipid ratio were observed in the ‘athletes’ performing prolonged exercise, compared with the controls. The researchers concluded that, in elite athletes, the integrity of the blood-gas barrier is altered only at extreme levels of exercise.
3. In Snyder, (2006) 15 min of ‘hypoxic’ exercise was performed at a workload that was initially equal to 40% of their maximal wattage performed during normoxia. BORG levels (at 5 mins) of levels ≤12 were considered low and resulted in an increase in wattage to 50% of normoxic max effort. This indicates an approximatuoion of latate threshold that may be defind as intense exercise even though not beyond lactate threshold.

The development of clinical pulmonary oedema increases remarkably reaching a 65% incidence rate when performing high intensity exercise (fig, 2), specifically when maximizing intensity and duration of exercise. Any bout of exercise that is submaximal, not of sufficient duration, and not to exhaustion is 4 times less likely to trigger interstitial pulmonary oedema, and even less likely to trigger alveolar oedema (Zavorsky, 2007). A significant deliniation in the broader category of edemic level exercise though subjective, is that subjects perceived effort was considered quite high, the general attitude being highly competitive and aimed at finishing efforts in the fastest possible time (Zavorsky, 2007). Also of note, the likelihood of developing pulmonary edema appeared to be independent of sex and ftness level.
These investigations provide a moderately clear demarkation between edemic and non edemic contextual variables except in the middle areas of moderate intensity with longer duration as not all the data is clear cut. Another good example of this opaic middle ground is illustrated by Burnham et al. (2009) who found data consistent with interstitial pulmonary edema with athletic subjects performing a 45 min cycle exercise at ∼70% VO2max. This may be considered sub lactate threshold (just). Thus it seems efforts need to be managed with care particularly when intensities approach very long duration, exaustion and or lactate inflection points.
The use of lower intensities for sustained efforts also seem to hold true at lower serum oxygen saturation levels. Hodges et al., (2007) found that transient interstitial pulmonary edema did not occur following sustained steady-state cycling exercise in normoxia or hypoxia (hypoxia of 15% O2), indicating that transient edema does not result from pulmonary capillary leakage during sustained submaximal exercise. This is significant as one of the symptoms of
chronic stage ARDS physiology is a sustained serum hypoxic status.
Diffusion Rate Adaption
In addition to acute exercise diffution rates, increases in exercise diffusion capacity via exercise adaption has been observed. Tedjasaputra (2016) describes an increase in membrane diffusion capacity and with no observed increases in pulmonary blood capillary volume. Increases in lung capillarization (cappilary numbers) has not been scientifically observed as a function of exercise or altitude adaption (Frisancho, 2013) . However, global cappilliarization and increases in haemoglibin concentrations are aerobic exercise adaptions generally. Increased haemoglobic has obvious benefits regarding the transport of oxygen to and from the lungs and in turn functional
capacity of the organism, while both haemoglobin and capilliarisation benefit other exercise specific organs such as muscle tissue.
Regarding diffusion capacity at the alveoli, there does appear to be intensity specific adaption changes as a result of exercise intensity. Dridi et al., (2021) showed that moderate intensity training (60% of HRmax) resulted in no change, while high intensity (70–80% of HRmax) showed improvements/increases in resting diffusion capacity ofter an 8 week trining progam. There were increases in Vc (lung capillary blood volume at rest), VO2max, TLNO (pulmonary diffusing capacity for nitric oxide) and DM (alveolar–capillary membrane diffusing capacity) in the IET (intense endurance training program) but not in the MET (moderate endurance training program) participants after eight weeks of training with varying exercise intensities.
Although the impact of increased defusion rate on edemic status was not evaluated, however one can infer that as adaption at higher intensity exercise improves, the rate of defusion and in turn oedem, may increase at rest and during subsequent higher intensity exercise efforts, thus increasing the risk of further lung consolidation.
Other Adaptations to Aerobic Endurance Training
Aerobic Fitness Mechanisms
Capacity to perform aerobic work, a combination of the maximal oxygen consumption (V̇O2 max), the product of cardiac output (CO) and arteriovenous oxygen (AV O2) difference at exhaustion, offers the possibility of selectively targeting individual components to boost aerobic power independently of rate of oxygen saturation (e.g. Cardioac Output). Factors affecting V̇O2 may also be classified as supply and demand; Firstly supply, which is the transport of
oxygen from the lungs (via alveoli) and then to the mitochondria (combining pulmonary function, cardiac output, blood volume, and the capillary density of the skeletal muscle). This is directly related to alveola defusion rates and is a limitation when concerning our current case. And secondly demand, which is the rate at which the mitochondria reduces oxygen in the process of oxidative phosphorylation and the generation of ATP. Mitochondrial efficiency may be
selectively targeted independently to boost functional capacity. Mitochondrial biosynthesis and efficiency enhances muscle oxidative capacity, supports greater generation of ATP, and in turn delays time to fatigue improving aerobic exercise performance (Margolis and Pasiakos, 2013).
Cardiac output, muscle capillarization and mitochondrial synthesis/function may be affected independently of the degree of O2 saturation (diffusion rate). Our question is, does this occur at sub maximal efforts?
Cardiac Output and Eccentric Cardiac Overload
Classic endurance training is known to result in enhanced cardiac output, maximal oxygen consumption, and mitochondrial biogenesis (Holloszy 1967; Coyle et al. 1983, 1986, 1988; Holloszy and Coyle 1984; Favier et al. 1986. (In Hughes et al., 2018)). Different exercise training protocols are thought to predominantly change cardiac workload either by pressure or by volume overload which may lead to different cardiac hypertrophy phenotypes. Aerobic exercise
training, such as long-distance running or swimming, is matched with an increased blood volume overload accompanied by cardiac chamber dilation, stimulating what is referred to as eccentric hypertrophy. This phenotype is associated with the addition of sarcomeres in series to lengthen the cardio-myocyte and to increase the width of the cell in parallel. Several studies in healthy subjects as well as in patients with heart failure (and reduced ejection fraction) have show improvements in maximal stroke volume (cardioac ouput) alongside VO2peak, and eccentric hypertrophy and conformed by MRI (Taylor et al., 2021; Spence et al., 2011). It should be noted that improvements are generally greater with high intensity training (50–70 % VO2peak) when compared with moderate intensity (Taylor et al., 2021).
The lower end of this intensity band (50-60% VO2max) allows some adaption to cardiac output capacity without exacerbation of edemic exchange into the alveoli.
Mitochondrail Function
As previously cited, Margolis and Pasiakos, (2013) discribe biosynthesis of mitochondria as enhancing skeletal muscle oxidative capacity, resulting in greater generation of ATP, and in turn delaying muscle time to fatigue, and while also improving aerobic exercise performance. Mitochondria from endurance trained muscles exhibit increases in the capacity to generate ATP via oxidative metabolism mediated by increases in the levels of the mitochondrial enzymes of the fatty acid oxidation pathway, citrate cycle, respiratory chain, and ATP synthesis. These findings have been confirmed in numerous studies on laboratory rodents and shown to also occur in humans (Holloszy, 2008). Increases in both the size and number of mitochondria are involved in this adaptive response and all of the muscle fiber types are involved in this adaptation (Holloszy, 2008).
Holloszy (2008) also describes how in addition to an increase in mitochondrial biogenesis/function, exercise induces an increase in the GLUT4 isoform of the glucose transporter. This increase in GLUT4 occurs in parallel with, and is mediated by the same signals and some of the same transcription factors as the increase in mitochondrial biogenesis. Of specific interaest to our investigation, GLUT4 is an insulin receptor and is relevant to glucose regulation and in
turn TGF-b whose it impact on lung diffution is discussed later in this discussion.
Mølmen et al., (2024) has reported on mitochondrial biogenesis using pooled data of over a hundred studies. While adjusting for covariates (intervention weeks, training frequency, initial fitness level, active muscle mass while training, disease status, sex, and age) with appropriate weighting, they found all training intensity categories display significant increases in mitochondrial content (ET, 22.7 ± 4.6%; HIT, 27.0 ± 5.1%; SIT, 27.0 ± 6.7%; all P < 0.001; model 3). Higher training frequencies (6 > 4 > 2 sessions/week) were also associated with larger increases in mitochondrial content. Endurance Training and High Intensity Training are defined as exercise training conducted at an intensity below or above the second ventilatory threshold/4 mmol/L blood lactate concentration/87% of HRmax/87% of ̇ VO2max/75% of Wmax (peak workload), respectively. Thus, sub lactate lower intensity endurance work should support mitochondrial adaption though no ‘minimum’ intensity threshold was established. A cursory review of the included research papers indicates a predominance of training protocols using exercise intensities at 70-75% VO2max by the final 4 weeks of training and with many protocols beginning at 50% of VO2max.
A Note on Sprint Training and Respiration Capacity
Numerous studies have found that short burst max effort training can improve metabolic function and fitness status. As an example, after 4 weeks of training, investigators observed a 25% increase in maximal mitochondrial respiration in the Sprint Interval Training (SIT) (30s sprints) (Hughes et al., 2018). In Granata et al., (2016), the maximal mitochondrial respiration in permeabilized muscle fibers increased significantly only after SIT (25%) [ SIT; 4-10 ×
30-s all-out bouts at ∼200% of peak power output (WPeak)]. Mølmen et al., (2024) also present similar results when analysing pooled data from multiple studies. Identically defined sprint intervals showed both mitochondrial biogenisis and increases in mitochondrial capacity.
Regarding the edemic capacity of short high intensity sprints, no direct data is available though data on blood pressure is. As a comparative baseline Nayor et al., (2022) describe changes from a resting systolic blood pressure of 120mm/Hg, peak systolic pressures of 180mm/Hg for continuous high intensity aerobic exercise. When compared to acute high intensity resistance training the differences are large. Palatini et al., (1989) saw extreme increases in BP
during heavy weight lifting at intensities of 90% of 1rm. For the squat, pressures of up tp 345/245mm/Hg was achieved within the working set, though pressures drop immediately at the end of efforts. However, during a handgrip to exhaustion challenge at 30% of 1rm an average increase of only 67mm/Hg was seen. Note, this is almost equivalent to continuous aerobic efforts. Santana et al., (2022) recorded blood pressure pre and post 10 x 30 meter all out
running sprint efforts with various rest intervals conditions of 20s, 30s or 40s. No statistical difference between pre and post exercise blood pressure values were found.
As incremental V̇O2 max tests not exceeding 20–25 min time have been shown not sufficient to trigger pulmonary edema (Miserocchi and Beretta, 2023; Zavorsky, 2007), and lower load higher rep sets to failure have been shown to mitigate large increases in blood pressure, one may infer short burst sprint intervals of 30-60 secs and multiple rep (e.g. 10-20) strength efforts are generally safer.
A note on ARDS and O2 Deffusion
Abnormal diffusion capacity testing, correlates with persistent imaging findings of fibrosis, muscular weakness, diminished mental state, and cognitive impairment following ARDS (see Fig 3). These are significant contributors to reduced exercise capacity and culminates in limited functional capacity and reduced quality of life. Exercise limitations may be related to muscular weakness, low serum O2 saturation and or comorbid cardiac disease. Exercise-induced oxygen desaturation below 88% suggesting ongoing pulmonary dysfunction is not uncommon (Farta and Warea, 2020), as is reduced oxygen transfer ( Neff et al., 2020).
Any reduction of muscle mass will translate as a reduction in functional capacity. Muscle mass is also a significant factor in the contribution to aerobic capacity, i.e. VO2max (Kim et al., 2016; Ozaki et al., 2013). Thus strength training may serve as a dual facilitator impacting both VO2max and relative strength.

Respiratory Muscles and Hypercapnea
Hypercapnia (increased blood CO2) usually coexists with decreased oxygen content and/or pH (acidosis). These conditions are capable of impairing respiratory muscle function which has been shown experimentally as depressed diaphragmic function (Nin et al., 2018). Peripheral muscles are generally not overloaded by hypercapnia, however if respiratory acidosis is induced due to the lungs inability to remove CO2, it can affect peripheral limb muscle
structure and function. Acute hypercapnia induced respiratory acidosis has been shown to depress limb muscle contractility and increase muscle fatigue induced by exercise in healthy subjects (Nin et al., 2018). Structural changes have been observed in the peripheral muscles of animals chronically exposed to CO2, although some findings are contradictory (Nin et al., 2018). Reduced intracellular pH is the most accepted mechanism for depressed muscle
contractility during hypercapnia (Schnader et al., 1985; Jarvis et al., 2018). Note that reduced pH also activates TGF-β which has a functional role in epithelial dysregulation (see section on TGF-β).
Of note, peripheral muscle strength and respiratory muscle function, are both requirements for effective O2 saturation and CO2 desaturation independently of current epithelial diffusion capacity. These deficits reduce ventilation via the reduction of respiratory muscle function, and peripheral muscles may effect posture (e.g. thoracic kyphosis), which can restrict lung inflation and in turn further restrict ventilatory capacity (Lorente et al., 2019).
Regarding exercise rehabilitation, Dunham and Harms (2012) has demonstrated significant improvements in respiratory muscle function with both High Intensity Interval Training and Moderate Intensity Training (43 vs. 25 %) also see Taylor et al., (2021) for a discussion. Respiratory muscles may also be trained independently of aerobic exercise training through the use of resisted breathing devices and will all likely contribute to improved ventilatory capacity (Winkle and Sankari, 2024).
Capilliarization and VEGF
Increased capillarisation is also a function of aerobic capacity. Mølmen et al., 2024 have shown that capillaries per muscle fiber increased similarly with Endurance Training (15 ± 3%), High Intensity Training (13 ± 4%) and Sprint Interval Training (10 ± 11%) (P = 0.556) after adjustments for number of intervention weeks and initial fitness level. However, Capillaries per mm2 only increased after ET (13 ± 3%) and HIT (7 ± 4%), with increases being larger after ET
compared with HIT and SIT (P < 0.05).
Vascular endothelial growth factor (VEGF) stimulates the formation of new blood vessels though there may be some discrepancies in the research on exercise intensity, VEGF and capillarisation. Many studies indicated that high- intensity intermittent exercise leads to lower VEGF levels and subsequently less endothelial cell proliferation compared with low-intensity exercise, with even evidence of a 22% reduction in VEGF levels following eight weeks of high-
intensity intermittent exercise (Mølmen et al., 2024). However, Wahl et al., (2014) has shown that VEGF significantly increased after HIT interventions as oposed to lower intensity high volume efforts.
Although the data on intensity, VEGF, and capilliarisation is not incontrovetible, one can assume that lower intensity endurance training may effectively increase capiulliarization to some degree and in turn improve aerobic capacity
and muscle function.
Exercise Prescription
Aerobic:
From the evidence presented it can be inferred that an aerobic base and with progressive increase in aerobic training volume is generally safer than high volume, high intensity training. 60-75% Max HR i(45-55% VO2max) is manageable at up to 15 mins and may be prolonged to 30 mins if monitored effectively and intensity remain low. Otherwise, one could perform 10mins at 50-60% maxHR, 15mins at 70+%, and then finally 5mins at 50-60%.
85% maxHR = (approx) 70% VO2max 100% maxHR = (approx) 100% VO2max
Power or shorter burst of anaerobic prescription should be limited and maintained at a low fixed volume. Continuous anaerobic and or interval training should be avoided for the duration of rehabilitation.
Example of a basic strategy:

Strength:
Strength training to accommodate increased muscle mass in the peripheral and core musculature and with some emphasis on posture is of benefit. The use of extended rest periods (3+ mins) will benefit the attenuation of metabolic build-up to maximise early adaption and minimise premature training fatigue. Sets should use larger rep ranges and an turn lower loads relative to 1rm.
E.g. 2 sets of 20 repetitions at 60% of 1rep max with 3mins recovery between sets.
Mechanical aids for improved ventilatory muscle tone may also be of benefit.
Drug Associated Interventions
Active Fluid Reabsorption (Na+ Transport)
Although Endogenous catecholamines are known to upregulate alveolar fluid clearance in several experimental models of acute lung injury, this upregulation may be short term and insufficient to
counterbalance excessive alveolar flooding (Matthay, 2002). Epithelial integrity is tightly related to the ability of the epithelial lining to function effectively. Injury to the alveolar epithelium can
disrupt the integrity of the alveolar barrier, increase rates of passive diffusion into the alveoli and downregulate active ion transport pathways, thus, reducing net alveolar fluid reabsorption and
in turn increasing the extent of alveolar oedema (Matthay, 2002).
Medical Drug Intervention/Physiologic Intervention
There are two plausible drug therapeutic pathways worth considering if complications occur.
Diuretics
Disruption of the normal oncotic pressure differential causes patients with ARDS to be particularly sensitive to the hydrostatic forces. Conservative fluid management, in which diuretics are
administered and intravenous fluid administration is minimized, may decrease hydrostatic pressure and increase serum oncotic pressure, potentially limiting the development of pulmonary
edema (Casey et al., 2019). Although this is primarily an acute mode therapeutic, diuretics for chronic management may also be worth considering during extended recovery periods. In addition to pharmacotherapeutics, fluid management and the use of caffeine or other organic diuretics can be considered.
B2-adrenergic agonists
B2-adrenergic agonists may influence a more sustained stimulation of alveolar fluid reabsorption. The positive, protective effects of β2-adrenergic receptor signalling on alveolar active Na+
transport in normal and injured lungs provide substantial support for the use of β-adrenergic agonists to accelerate alveolar fluid clearance in patients with cardiogenic and noncardiogenic
pulmonary edema (Gökhan and Factor 2008).
‘Activation of β2-adrenergic receptor (β2AR) increases alveolar active Na+ transport via upregulation of epithelial Na+ channel (ENaC) and cystic fibrosis transmembrane conductance regulator (CFTR) as well as basolaterally located Na,K-ATPase (open arrows). Activation of the receptor also increases β-catenin and surfactant release, which might be important in the
pathogenesis/resolution of acute lung injury’ (in, Gökhan and Factor 2008). β2AR agonists also have broad anti-inflammatory effects in the setting of ALI (Bosmann et al., 2012), which may reduce the impact of any residual inflammatory contribution.
In addition to the use of pharmaceutical agents, natural hormones also stimulate B2 receptors. Epinephrine (adrenaline) is the most effective natural catecholamine agonist of B2, while norepinephrine (noradrenaline) is less effective (Abosamak and Shahin, 2023).
Catecholamine stimulation of β2AR receptors can be achieved naturally through modalities such as sprint exercise (Zouhal et al., 2008), sauna (Podstawski, 2019) and deliberate cold exposure (Brigant, 2023).
Secondary Mechanisms of Epithelial Damage
Food Allergies/Intolerances
A Note on Lipopolysaccharide (LPS) (Endotoxins) and Epithelial Damage
Endotoxins, also known as lipopolysaccharides (LPS), are a major component of the outer membrane of Gram-negative bacteria (Omar, 2021). LPS are able to make a great impact on gut homeostasis as the colonic lumen contains a plethora Gram-negative bacteria that are capable of LPS release. A higher expression of endotoxin/LPS in the blood can indicate epithelial barrier dysfunction and promote inflammatory processes (Ghosh, 2020). LPS from bacterial
infection is a potent activator and even minute amounts in the blood is sufficient to induce potent inflammatory response (Rhee, 2014).
Commensal microbiota have been shown to contribute to the development of leaky gut syndrome (LGS) and (LGS)-associated autoimmune diseases (Kinashi and Hase, 2021). More significantly, serum levels of LPS have been shown to be significantly higher in such cases, as analysis of patients with Graves’ Disease as compared to healthy controls has shown (Zheng et al., 2021). This supports the hypothesis that serum LPS via intestinal permeability may
be able contribute to endothelial disruption at the lungs and in turn impact on ARDS recovery. This seems all the more plausible when one considers the following experimental evidence. Alveolar fluid has been demonstrated to accumulate in a mouse model of LPS-induced lung injury. But when VE-cadherin bonds are stabilized by genetic alterations that prevent breakdown or by blocking VE phosphodiesterase, there is reduced edema formation (Schulte, 2011; Broermann, 2011; Huppert, 2019). VE-cadherin plays a key role in the remodeling, gating, and maturation of vascular vessels and zonulin (see below) alters their expression by mechanisms that include downregulation of VE- cadherin (Ciccia et al., 2017). Zonulin is a key player in intestinal permeability.
Wheat/Zonulin and Leaky Lungs
Gluten, and potentially other gluten mimicking compounds have been established as a causal factor in leaky gut (intestinal endothelial permeability). This is a direct result of wheat
increasing zonulin concentrations. When the zonulin pathway is deregulated, intestinal and extraintestinal autoimmune, inflammatory, and neoplastic disorders can occur in
genetically susceptible individuals (Fasano, 2011). Zonulin has also been shown to modulate human bronchial epithelial barrier function, and shows that serum zonulin has the potential
to modulate function at the bronchoalveolar lining (Romem, 2012).
In an experimentally induced epithelial injury, Rittirsch et al., (2012) used confocal microscopy (laser scanning) to examine sections of injured mouse lungs, and observed discontinuous, reduced and fragmented epithelial tight junctions. This included blood products leaking into the alveoli as a result of bacterial lipopolysaccharide (LPS) injured lungs. Application of the Zonulin agonist AT-1002 intensified the Lung injury. Blockade of Zonulin resulted in an attenuation not only of lung permeability but also the extent of pulmonary polymorphonuclear neutrophil (PMN) accumulation and the appearance of inflammatory cytokines in BAL fluids (a diagnostic method). This indicates that Zonulin may be involved in events during the pathogenesis and recovery of acute lung injury. Note, PMN is a defensive phagocyte which is often necessary to clear bacteria from the lung.
As zonulin may be a significant contributor to epithelial dysfunction at the lungs, dietary modification may be an avenue for modulation of blood serum zonulin levels and in turn lung epithelial status. And indeed, the presence and modulation of blood serum zonulin via dietary intervention has been experimentally observed. In a six-month wheat avoidance strategy, Barbaro et al., (2020) showed significantly reduced blood serum zonulin levels in HLA-DQ2/8-positive participants with non-Celiac Gluten Sensitivity. This indicates that at least in gluten sensitive individuals, systemic circulation of Zonulin may be present in the blood at elevated levels (gluten sensitive participants had higher zonulin levels) and may be reduced through dietary restriction of gluten/wheat.
Sugars and Zonulin
Fructose/Galactose malabsorption promotes gut permeability, and higher fructose intake promotes gut inflammation, an accompanying rise in blood endotoxin (LPS) concentrations due to endothelial dysfunction, and a decline of tight-junction proteins associated with Zonulin levels (Lambertz et al., 2017; Volynets et al., 2017; Binienda et al., 2020). In animal
models, Omar et al., (2021) has shown the potential adverse effects of high galactose and fructose intake on metabolic factors, and gut integrity markers independently of any
malabsorption scenario. However, inconsistent changes in zonulin concentrations were observed in response to endotoxin expression in the plasma.
Of further interest, Ohlsson et al., (2017) found that higher zonulin levels were associated with higher waist circumference, diastolic blood pressure, glucose levels, being overweight, obesity, and hyperlipidemia. However, zonulin levels among individuals reporting GI symptoms or GI diseases were not correlated.
Although research on the impact of sugars on zonulin concentrations is somewhat opaque with regards to our question on alveoli integrity, we may assume that excess levels may
induce leaky gut and inflammatory reactions in the body, and that there is a potential for increased blood serum zonulin concentration especially in over weight and insulin resistant individuals.
it should be noted that in addition to carbohydrates, lipid composition has been shown to affect intestinal permeability. Free fatty acids seem to have a beneficial effect on gut permeability, for instance SCFAs, mainly butyrate may lead to the reduction of intestinal permeability (Binienda et al., 2020; Leonel and Alvarez-Leite, 2012).
Food Considerations
With regards to the data on Zonulin and sugars, a limitation/Management of dietary wheat/gluten (and gluten mimicking substances). Sugars and including lactose, may also be an
effective adjunct to rehabilitation. The Paleo diet is ideal as a general conceptual framework which should also encourage ample natural source essential fatty acids.
Any specific food intolerance and malabsorption issues should also be addressed.
Inflammation and Metabolic Regulation
Herrero et al., (2017) describes a broad array of factors that may contribute to epithelial damage and the resultant edematic impact on the alveoli of the lungs. This includes dysregulated
inflammation, leukocyte infiltration, activation of pro-coagulants, apoptosis and mechanical stretch. Below is a brief overview of some of the mechanisms associated with this
type of endothelial disruption, and some plausible interventions.
TGF-β
Transforming growth factor beta (TGF-β) is a multifunctional cytokine. It is related to integrins, obesity, acids, bases and Thrombospondin-1. TGF‐β is secreted in an inactive (latent) form in a complex with two proteins - LAP and LTBP. Its activation requires the dissociation from the complex, which occurs at a low pH, through the action of reactive oxygen species, proteases, thrombospondin 1 and or several integrins (Margadant & Sonnenberg, 2010).
TGF-β is in part responsible for the physiological disruption associated with insulin resistance and type 2 diabetes (Lee et al., 2021) (also see glycation and insulin section). For example, in one study, insulin resistant transgenic mice overexpressing TGF-β1 were shown to exhibit fibrosis and increased fatty deposits at the pancreas, while TNF-α, a known inflammatory marker related to both
fibrosis and autoimmunity dramatically decreased the number of insulin-containing beta cells (Sanvito, 1995). With regards to alveola edema, TGF-β can block active Na+ transport at the epithelium and reduce alveola fluid clearance (Peters et el., 2013).
Of note, aerobic exercise may improve TGF-b status (Wen et al., 2022) and simultaneously improve insulin resistance, while respiratory acidosis due to increased serum CO2 may be counterproductive.
Integrin Activation of TGF-β
Integrins are transmembrane receptors that help cell-cell and cell-extracellular matrix (ECM) adhesion. They play pivotal roles that are crucial to infection, inflammation and
angiogenesis. Other functions include cell proliferation, migration, apoptosis, and tissue repair. The activity of integrins depend on numerous factors. These factors can be either
activators or inhibitors. Factors include hormones, cytokines, mediators of systemic inflammation, active components of the complement system, active oxygen species, endotoxins and or pharmacological compounds (Mrugacz et al., 2021).
‘Integrin-dependent’ activation of TGF-β is so far the best described and likely the most important mechanism ascribed to integrins. TGF-β activation mediated by acids, bases, reactive oxygen
species (ROS), thrombospondin-1 (TSP-1), proteases, and other TGF-β activators is collectively known as ‘integrin-independent’ activation (Deng et al., 2024). Among all integrins, αVβ6 and αVβ8 integrins are the best studied TGF-β activators. The lack of αVβ6 and αVβ8 integrin activity reproduces the phenotypes of TGF-β1- and TGF-β3-null mice, indicating the central importance of integrin-dependent activation Deng et al., (2024).
Obstruction of integrin-dependent TGF-β activation is seen as a promising remedy for certain inflammatory diseases and allergic inflammatory reactions (Kudo et al., 2012; Nolte
and Margadant, 2019). Of note in lung inflammation, mice lacking the αvβ8 integrin on dendritic cells do not generate TH17 cells (inflammatory cells) in the lungs. They are
protected from airway hyper-reactivity in response to house dust mites, and ovalbumin sensitization and challenge (Kudo et al., 2012).
Dietary Factors and Disease
Fenton et al., (2016) have shown that the key activator of TGF-β, integrin αvβ8, is significantly upregulated on the intestinal Dendritic Cells (DC) of Inflammatory Bowel Disease (IBD) patients,
indicating that inflammatory signals may upregulate expression of this key TGF-β -activating molecule. In fact it was found that the Toll-like receptor 4 ligand lipopolysaccharide upregulates
integrin avb8 expression and TGF-β activation by human DC (also see section: A Note on Lipopolysaccharide (LPS)/Endotoxins). The researchers concluded that microbial signals enhance the TGF-β activating ability of human DCs via regulation of integrin αvβ8 expression. Thus, intestinal inflammation may drive this pathway in patients with IBD, a proposition also congruent with food intolerance reactions and local allergic responses (Ha et al., 2021; Pasta et al., 2024; Farhana and Khan, 2023; Dlugosz et al., 2015).
Blood serum levels including TNF-α and TGF-β are elevated in ulcerative colitis patients (Feng et al., 2014), TGF-β elevated in allergy (Weissler and Frischmeyer-Guerrerio, 2019), and hsCRP elevated in Lactose Intolerance and independently of Casein content (Ramakrishnan et al., 2024). Moraes et al., (2020) has shown that serum inflammatory markers are not elevated in Inflammatory Bowel Syndrome (IBS) when compared to norms.
This pathway highlights a possible strategy to aid the minimisation of lung edema through TGF-β reduction via effective respiratory allergen management, management of nutritional allergen
sensitivities more broadly, food intolerance and the effective management of autoimmunity.
A Note on Thrombospondin 1 (TSP1)
Thrombospondin 1 (TSP1) regulates innate and adaptive immune responses through the transmembrane proteins CD47, CD36 and CD47. It also mediates cell-to-cell and cell-to-matrix interactions. There is a prevalence of TSP1 in inflammation and obesity (Gutierrez and Gutierrez, 2021) and under hyperglycaemic conditions TSP1 expression is enhanced and results in
upregulation of TSP1 gene expression (Gutierrez and Gutierrez, 2021). Studies have demonstrated a regulatory role for (TSP1) in cell migration, and in activation of the latent
transforming growth factor beta 1 (TGF-β1) as a function of metabolic dysregulation (Gutierrez and Gutierrez, 2021).
TSP1 also enhances vasoconstriction and reduces local blood flow by blocking the function of nitric oxide’s ability to relax vascular smooth muscle (Kaur and Roberts, 2023).
Increased vasoconstriction increases blood/hydrostatic pressure and can contribute to the likelihood of pulmonary edema. It may also reduce the rate of Oxygen and
CO2 exchange under certain conditions. Under normal circumstances, regional alveolar hypoxia causes local vasoconstriction at the lung, shifting blood flow from hypoxic to normoxic areas, thereby helping to optimise gas exchange. Acute disturbances in this mechanism can be life-threatening and chronic hypoxia will trigger lung vascular remodelling and pulmonary hypertension
(Weissmann et al., 2006). This mechanism is known as hypoxic pulmonary vasoconstriction (HPV) and involves biological oxygen sensing, pulmonary arterial smooth muscle cell contraction, and
modulation of these responses (Tarry and Powell, 2017).
Thus, as a by-product of hyperglycaemia and obesity, TSP1 my contribute to delayed recovery via exaggerated vasoconstriction and TGF-β1 activation.
Chronic Glycation and Insulin Resistance
The inflammatory response as a result of hyperglycemia and insulin resistance could be a major factor for the increased expression in TGF-β1. In women with prior history of gestational diabetes mellitus (pGDM), Yener et al., (2006) using multiple regression analysis, demonstrated elevated TGF-β1 levels in pGDM as a function of post prandial glucose, age and BMI. This is may indicate that chronic long term glucose exposure is a significant factor and in addition to acute postprandial status. This analysis also showed that post prandial glucose, age and BMI were important correlates to TSP1 (Yener et al., 2006). That is, increases in TSP1 (TSP1 regulates nitric oxide and activates latent TGF-β1) were also shown in these women. In children with type 1 Diabetes Mellitus (T1DM), glycation, oxidation and TGF-β were higher than controls. TGF-β also correlated with age and duration of T1DM (Jakus et al., 2012). However, in this study, there was no significant relation between TGF-β and acute parameters of glycation and oxidation. This may highlight that metabolic dysregulation is a covariable as T1DM is purely an autoimmune
condition whereas T2DM occurs as a function of chronic metabolic issues.
In an interesting study of serum glucose concentrations by Pscherer et al., (2013) showed that control subjects had higher levels of TGF-β than DPP-4 inhibitor treated type 2 diabetics (DPP-4
inhibitors reduce glucagon and blood glucose levels). The researchers hypothesised that the blood glucose impact on TGF-β may be independent of diagnostic status, and that ‘norms’ may benefit from blood glucose reduction strategies. It should be noted that T2DM is medically diagnosed purely by serum glucose levels and independent of metabolic status.

Other studies have also reported the associations of TGF-β with blood glucose regulation. Plasma levels of TGF-β are elevated in non-insulin dependent Diabetes Melitus (Wen et al., 2022), and TGF-β seems corelated with chronic insulin resistance, glycation and oxidative stress (Lee et al., 2021).
Chronic exposure to hyperglycemia also leads to the accumulation of reactive oxygen species (ROS) activating damaging pathways which trigger nitro-oxidative stress (NOS) and
endoplasmic reticulum stress, Robertson (2004); Lee et al., (2021). These pathways are known drivers of TGF-β activation.
Alcohol
Ethanol by itself does not stimulate insulin secretion, however it does have an antagonistic effect on insulin receptor affinity. Metz et al., (1969) showed that pre-treatment with ethanol augments the plasma insulin response in the presence of glucose. Another study supported that ethanol had no effect on basal, but significantly enhanced glucose-stimulated
insulin concentrations (Shelmet et al., 1988), see Fig 5.
Though there is an absence of an increased glucose response to pure ethanol, and no change to overall glucose load in the presence of ethanol, two main points of interest are worthy of note. One, alcohol is rarely consumed outside of any accompanying carbohydrate mixture, and secondly TGF-β is independently stimulated by alcohol (Gerjevic et a;., 2012).
Jogunoori and Mishra (2018) describe how acute alcohol exposure induces TGF-β signalling, and upon chronic exposure, TGF-β illicits a pro-fibrotic response through TGF-β mediated activation of p38-MAPK signalling. This signalling process is directly associated with steatosis, cirrhosis and eventual hepatocellular carcinoma.

Dietary Intervention
Reduction in glucose load and minimal alcohol consumption offer further strategic reductions in epithelial barrier function optimisation
for active fluid transport.
Continuous glucose monitoring with an aim to target food spikes, Paleo Diet, Low Carb and alcohol moderation with and aim to reduce
weight or avoid weight gain.
References:
Abosamak, N. and Shahin, M. (2023) ‘Beta2 Receptor Agonists and Antagonists’, [Updated 2023 Jul 3]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2024 Jan-. Available from:
https://www.ncbi.nlm.nih.gov/books/NBK559069/ PMID: 32644495
Barbaro, M., Cremon, C., Morselli-Labate, A., Di Sabatino, A., Giuffrida, P., Corazza, G., Di Stefano, M., Caio, G., Latella, G., Ciacci, C., Fuschi, D., Mastroroberto, M., Bellacosa, L., Stanghellini, V., Volta,U. and Barbara, G. (2020)
‘Serum zonulin and its diagnostic performance in noncoeliac gluten sensitivity’, Gut, Vol. 69: pp1966–1974. doi:10.1136/gutjnl-2019-319281
Bates, M., Farrell, E., and Eldridge, M. (2011) ‘The Curious Question of Exercise-Induced Pulmonary Edema’ Pulm Med, Vol.2011: pp2011. doi: 10.1155/2011/361931
Binienda, A., Twardowska, A., Makaro, A., Salaga, M., (2020) ‘Dietary Carbohydrates and Lipids in the Pathogenesis of Leaky Gut Syndrome: An Overview’, Int J Mol Sci, Vol. 8;(21): pp8368. doi: 10.3390/ijms21218368
Bosmann, M., Grailer, J., Zhu, K., Matthay,M., Sarma, J., Zetoune, F. and Ward, P. (2012) ‘Anti-inflammatory effects of β2 adrenergic receptor agonists in experimental acute lung injury’, Faseb J. Vol. 26(5): pp2137–2144. doi:
10.1096/fj.11-201640
Briganti, G., Chesini, G., Tarditi, D., Serli, D. and Capodici, A. (2023) ‘Effects of cold water exposure on stress, cardiovascular, and psychological variables’, Acta Physiologica, Vol. 239(e14056): DOI: 10.1111/apha.14056
Broermann, A., Winderlich, M., Block, H., Frye, M., Rossaint, J., Zarbock, A., Cagna, G., Linnepe, R., Schulte,D., Nottebaum, A., and Vestweber, D., (2011) ‘Dissociation of VE PTP from VE-cadherin is required for leukocyte
extravasation and for VEGF-induced vascular permeability in vivo’, J Exp Med, Vol. 208(12): pp2393–2401. doi: 10.1084/jem.20110525
Burnham, K., Arai, T., Dubowitz, D., Henderson, A., Holverda, S., R. B. Buxton, R., Prisk, G. and S. R. Hopkins, S. (2009) ‘Pulmonary perfusion heterogeneity is increased by sustained, heavy exercise in humans’, J Appl Physiol, Vol.
107(5):1pp559-68. doi: 10.1152/japplphysiol.00491.2009.
Casey, J., Semler, M., and Rice, T. ( 2019) ‘Fluid Management in ARDS’, Semin Respir Crit Care Med, Vol. 40(1): pp57–65. doi:10.1055/s-0039-1685206.
Ciccia, F., Guggino, G., Rizzo, A., Alessandro, R., Luchetti, M., Milling, S., Saieva, L., Cypers, H., Stampone, T., Di Benedetto, P., Gabrielli, A., Fasano, A., Elewaut, D. and Triolo, G. (2017) ‘Dysbiosis and zonulin upregulation alter gut
epithelial and vascular barriers in patients with ankylosing spondylitis’’, Ann Rheum Dis, Vol. 6(6): pp1123–1132. doi:10.1136/annrheumdis-2016-210000.
Deng, Z., Fan, T., Xiao, C. Tian, H., Zheng, Y., Li, C. and He, J. (2022) ‘TGF-β signaling in health, disease, and therapeutics’, Signal Transduct Target Ther, Vol. 9(1): pp61. doi: 10.1038/s41392-024-01764-w.
Dlugosz, A., Nowak, P., D’amato, M., Kermani, G., Nystrom, J., Abdurahman, A. and Lindberg, G. (2015) ‘Increased serum levels of lipopolysaccharide and antiflagellin antibodies
in patients with diarrhea predominant irritable bowel syndrome’, Neurogastroenterol Motil, Vol. 27: pp1747–1754. doi: 10.1111/nmo.12670
Dridi, R., Dridi, N., Govindasamy, K., Gmada, N., Aouadi, R., Guénard, H., Laher, I., Saeidi, A., Suzuki, K., Hackney, A. and Zouhal, H. (2021) ‘Effects of Endurance Training Intensity on Pulmonary Diffusing Capacity at Rest and after
Maximal Aerobic Exercise in Young Athletes’, Int. J. Environ. Res. Public Health, Vol. 2021(18): 12359. https://doi.org/10.3390/ijerph182312359
Dunham, C. and Harms, C. (2012) ‘Effects of high-intensity interval training on pulmonary function’, Eur J Appl Physiol, Vol. 112: pp3061–8. doi: 10.1007/s00421-011-2285-5
Effros, R. and Parker, J. (2009) ‘Pulmonary vascular heterogeneity and the Starling hypothesis’ Microvasc Res, Vol. 78(1): pp71–77. doi: 10.1016/j.mvr.2009.03.004.
Farhana A, Khan YS. Biochemistry, Lipopolysaccharide. [Updated 2023 Apr 17]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2024 Jan-. Available from:
Fasano, A. (2011) ‘Zonulin and Its Regulation of Intestinal Barrier Function: The Biological Door to Inflammation, Autoimmunity, and Cancer’, Physiol Rev, Vol. 91(1): pp151–175. doi:10.1152/physrev.00003.2008
Feng, J., Yang, Z., Zhu, Y., Liu, Z., Guo, C. and Zheng, X. (2014) ’Serum IL-17 and IL-6 increased accompany with TGF-β and IL-13 respectively in ulcerative colitis patients’, Int J Clin Exp Med, Vol. 7(12): pp5498–5504. https://pmc.ncbi.nlm.nih.gov/articles/PMC4307508/
Fenton, T., Kelly, A., Shuttleworth, E., Smedley, C., Atakilit, A., Powrie, F., Campbell, S., Nishimura, S., Sheppard, D., Levison, S., Worthington, J., MJ Lehtinen, M. and Travis, M. (2017)
‘Inflammatory cues enhance TGFb activation by distinct subsets of human intestinal dendritic cells via integrin avb8’, Mucosal Immunol, Vol.10(3): pp624-634. doi: 10.1038/mi.2016.94. Epub
2016 Oct 26.
Fermoyle, C., Stewart, G., Borlaug, B. and Johnson, B. (2020) ‘Effects of exercise on thoracic blood volumes, lung fluid accumulation, and pulmonary diffusing capacity in heart failure with preserved ejection fraction’, Am J Physiol
Regul Integr Comp Physiol, Vol. 319: R602–R609. doi:10.1152/ajpregu.00192.2020.
Frank, J and Matthay, M. (2014) ‘TGF-β and lung fluid balance in ARDS’, Proc Natl Acad Sci U S A, Vol.111(3):pp885-6. doi: 10.1073/pnas.1322478111.
Frisancho, R. (2013) ‘Developmental Functional Adaptation to High Altitude: Review’, Am J Hum Biol, Vol. 25(2): pp151-68. doi: 10.1002/ajhb.22367.
Gerjevic, L., Liu, N., Lu, S. and Harrison-Findik, D. (2011) ‘Alcohol Activates TGF-Beta but Inhibits BMP Receptor-Mediated Smad Signaling and Smad4 Binding to Hepcidin Promoter in the Liver’, Int J Hepatol, Vol. 2012(2012): 459278.
doi: 10.1155/2012/459278.
Ghosh, S., Wang, J., Yannie, P. and Ghosh, S. (2020) ‘Intestinal Barrier Dysfunction, LPS Translocation, and Disease Development’, J Endocr Soc, Vol. 4(2): bvz039. doi: 10.1210/jendso/bvz039
Giuseppe Miserocchi, G. and Beretta, E. (2023) ‘A century of exercise physiology: lung fluid balance during and following exercise’ European Journal of Applied Physiology, Vol. 123: pp1–24. https://doi.org/10.1007/s00421-022-05066-3
Granata, C., Oliveira, R., Little, J., Renner, K. and Bishop, D. (2016) ’ Training intensity modulates changes in PGC-1α and p53 protein content and mitochondrial respiration, but not markers of mitochondrial content in human skeletal
muscle’, FASEB J, Vol. 30(2): pp959-970. doi: 10.1096/fj.15-276907.
Gutierrez, L. and Gutierrez, J. (2021) ‘Thrombospondin 1 in Metabolic Diseases’, Front Endocrinol (Lausanne). Vol.12: 638536. doi: 10.3389/fendo.2021.638536
Ha, E., Kim, J.,Yon, D., Lee, S.,Kim, M.,Lee, K., Sung, M., Jee, H., Shin, Y. and Han, H. (2021) ‘Association of serum lipopolysaccharide-binding protein level with sensitization to food
allergens in Children’, Sci Rep, Vol. 11(1): pp2143. doi: 10.1038/s41598-020-79241-x.
Herrero, R., Sanchez, G., and Lorente, J., (2017) ‘New insights into the mechanisms of pulmonary edema in acute lung injury’, Ann Transl Med, Vol 6(2): 32. doi: 10.21037/atm.2017.12.18
Hodges, A., Sheel, W., Mayo, J. and McKenzie, D. (2007) ‘Human lung density is not altered following normoxic and hypoxic moderate-intensity exercise: implications for transient edema’, J Appl Physiol, Vol. 103: pp111–118.
doi:10.1152/japplphysiol.01087.2006.
Hodges, N., Mayo, J. & McKenzie, D. (2005) ‘Sustained exercise does not alter lung density in humans’, Can J Appl Physiol 30 (Suppl.), S37.
Hughes,D., Stian Ellefsen, S. and Baar, K. (2018) ‘Adaptations to Endurance and Strength Training’, Cold Spring Harb Perspect Med, Vol. 8(6): a029769. doi: 10.1101/cshperspect.a029769
Holloszy, J. (2008) ‘Regulation by exercise of skeletal muscle content of mitochondria and GLUT4’, Review J Physiol Pharmacol, Vol. 59(7): pp5-18. PMID: 19258654
Huppert, L., Matthay, M., and Ware, L. (2019) ‘Pathogenesis of Acute Respiratory Distress Syndrome’, Semin Respir Crit Care Med, Vol. 40(1): pp31–39. DOI https://doi.org/10.1055/s-0039-1683996.
Jaku, V., Sapak, M., and Kostolanska, J. (2012) ‘Circulating TGF-β1, Glycation, and Oxidation in Children with Diabetes Mellitus Type 1’, Exp Diabetes Res, Vol. 2012 (Article ID 510902): 7 pages. doi:10.1155/2012/510902
Jarvis, K., Woodward, M., Debold, E. and Walcott, S. (2018) ‘Acidosis affects muscle contraction by slowing the rates myosin attaches to and detaches from actin’, J Muscle Res Cell Motil, Vol. 39(3-4): pp135-147. doi: 10.1007/s10974-
018-9499-7. Epub 2018 Oct 31.
Jogunoori, W. and Mishra, L. 2018. Role of TGF-β in Alcohol-Induced Liver Disease. In Vasiliou, V. et al., eds. Alcohol and Cancer. Adv Exp Med Biol, Vol. 1032: pp93-104. doi: 10.1007/978-3-319-98788-0_7.
Kim, C., Courtney M. Wheatley, C., Behnia, M. and Bruce D. Johnson. B. (2016) ‘The Effect of Aging on Relationships between Lean Body Mass and VO2max in Rowers’ PLoS ONE, Vol. 11(8), pp1-11. Article e0160275.
Kinashi, Y. and Hase, K. (2021) ‘Partners in Leaky Gut Syndrome: Intestinal Dysbiosis and Autoimmunity’, Front Immunol, Vol.12:673708. doi: 10.3389/fimmu.2021.673708
Kudo, M., Melton, A., Chen, C., Engler, M., Huang, K., Ren, X., Wang, Y., Bernstein, X., Li, J., Atabai, K., Huang, X. and Sheppard, D. (2012) ‘IL-17A produced by αβ T cells drives airway hyper-responsiveness in mice and enhances mouse
and human airway smooth muscle contraction’, Nat Med, Vol. 18(4): pp547–554. doi:10.1038/nm.2684
Lambertz, J., Weiskirchen, S., Landert, S., and Weiskirchen, R. (2017) ‘Fructose: A Dietary Sugar in Crosstalk with Microbiota Contributing to the Development and Progression of Non-Alcoholic Liver Disease’, Front. Immunol, Vol. 8:
pp1159. doi: 10.3389/fimmu.2017.01159
Lee, J., Lee, J. and Rane, S. (2021) ‘TGF-β Signaling in Pancreatic Islet β Cell Development and Function’, Endocrinology, 2021, Vol. 162(3): pp1–10. doi:10.1210/endocr/bqaa233
Leonel, A. and Alvarez-Leite, J. (20212) ‘Butyrate: implications for intestinal function’, Curr Opin Clin Nutr Metab Care, Vol. 15(5): pp474-9. doi: 10.1097/MCO.0b013e32835665fa.
Lorente, A., Barrios, C., Lorente, R., Tamariz, R. and Burgos, J. (2019) ‘Severe hyperkyphosis reduces the aerobic capacity and maximal exercise tolerance in patients with Scheuermann disease’, Spine J, Vol. 19(2): pp330-338. doi:
10.1016/j.spinee.2018.07.002. Epub 2018 Jul 17.
Manier, G., Duclos, M., Arsac, L., Moinard, J. & Laurent, F. (1999) ‘Distribution of lung density after strenuous, prolonged exercise’, J Appl Physiol, Vol. 87(1): pp83-9. doi: 10.1152/jappl.1999.87.1.83.
Margadant, C. & Sonnenberg, A. (2010) ‘Integrin–TGF-β crosstalk in fibrosis, cancer and wound healing’, EMBO reports, Vol. 11(2): pp97–105. doi: 10.1038/embor.2009.276
Margolis, L and Pasiakos, S. (2013) ‘Optimizing Intramuscular Adaptations to Aerobic Exercise: Effects of Carbohydrate Restriction and Protein Supplementation on Mitochondrial Biogenesis‘ Adv. Nutr. Vol. 4: pp657–664.
doi:10.3945/an.113.004572.
Mart, M. and Ware, L. (2020) ‘The long-lasting effects of the acute respiratory distress syndrome’, Expert Rev Respir Med, Vol. 14(6): pp577–586. doi:10.1080/17476348.2020.1743182.
Metz, R., Berger, S., and Mako, M, (1969) ‘Potentiation of the Plasma Insulin Response to Glucose by Prior Administration of Alcohol An Apparent Islet-Priming Effect’, Diabetes, Vol. 18(8): pp517–522.
Miserocchi, G. and Beretta, E. (2023) ‘A century of exercise physiology: lung fuid balance during and following exercise’, European Journal of Applied Physiology, Vol.123(1): pp1–24.
Mølmen, K., and Almquist, N. and Skattebo, Ø. (2024) ‘Effects of Exercise Training on Mitochondrial and Capillary Growth in Human Skeletal Muscle: A Systematic Review and Meta‑Regression’, Sports Med. 2024 Oct 10. doi:
10.1007/s40279-024-02120-2. Epub ahead of print. PMID: 39390310.
Moraes, L., Magnusson, M., Mavroudis, G., Polster, A., Jonefjäll, B., Törnblom, H., Sundin, J., Simrén, M., Strid, H. and Öhman, L. (2020) ‘Systemic Inflammatory Protein Profiles Distinguish Irritable Bowel Syndrome (IBS) and Ulcerative
Colitis, Irrespective of Inflammation or IBS-Like Symptoms’, Inflamm Bowel Dis, Vol. 26(6): pp874-884. doi: 10.1093/ibd/izz322.
Mrugacz, M., Bryl, A., Falkowski, M., and Zorena, K. (2021) ‘Integrins: An Important Link between Angiogenesis, Inflammation and Eye Diseases’, Cells, Vol. 10(7): pp1703. doi: 10.3390/cells10071703.
Mutlu, G. and Factor, P. (2008) ‘Alveolar Epithelial β2-Adrenergic Receptors’, Am J Respir Cell Mol Biol, Vol. 38(2): pp. 127–134. doi: [online] 10.1165/rcmb.2007-0198TR
Nayor, M., Gajjar, P., Murthy, V.,Vasan , R., Lewis, G., Miller, P., Velagaleti, R., Larson, M., Mitchell, G. and Shah, R. (2023) ‘Blood Pressure Responses During Exercise: Physiological Correlates and Clinical Implications’, Arterioscler
Thromb Vasc Biol, Vol. 43: pp163–173. DOI: 10.1161/ATVBAHA.122.318512
Neff, T., Stocker, R. Frey, H., Stein, S. and Russi, E. (2003) ‘Long-term assessment of lung function in survivors of severe ARDS’, Chest, Vol. 123(3): pp845–853. doi: 10.1378/chest.123.3.845.
Nin, N., Angulo, M. and Briva, A. (2018) ‘Effects of hypercapnia in acute respiratory distress syndrome’, Ann Transl Med, Vol. 6(2): pp37. doi: 10.21037/atm.2018.01.09
Nolte, M. and Coert Margadant, C. (2019) ‘Controlling Immunity and Inflammation through Integrin-Dependent Regulation of TGF-b’, Trends Cell Biol, Vol. 30(1): pp49-59. doi: 10.1016/j.tcb.2019.10.002
O’Brodovich, H. (2001) ‘Pulmonary edema fluid movement within the lung’, Am J Physiol Lung Cell Mol Physiol, Vol. 281(6): pp1324–L1326. doi: 10.1152/ajplung.2001.281.6.L1324.
Ohlsson, B., Orho-Melander, M., Peter M Nilsson., P. and Angelini, S (ed) (2017) ‘Higher Levels of Serum Zonulin May Rather Be Associated with Increased Risk of Obesity and Hyperlipidemia, Than with Gastrointestinal Symptoms or
Disease Manifestations’, Int J Mol Sci, Vol.18(3): pp582. doi: 10.3390/ijms18030582
Omar, N., Kruger, J., Bello, F., Medana, C., Collino, M., Zamaratskaia, G., Michaelsson, K., Wolk, A., and Landberg, R. (2021) ‘Effects of High Intakes of Fructose and Galactose, with or without Added Fructooligosaccharides, on
Metabolic Factors, Inflammation, and Gut Integrity in a Rat Model’, Mol. Nutr. Food Res, Vol. 65(6): pp2001133. DOI: 10.1002/mnfr.202001133
Ozaki, H., Loenneke, J., Thiebaud, R. & Takashi Abe, T. (2012) ‘Resistance training induced increase in VO2max in young and older subjects’, Eur Rev Aging Phys Act, Vol. 10: pp107–116. DOI 10.1007/s11556-013-0120-1
Palatini, P., Mos, L., Munari, L.,Valle, F., Del Torre, M., Rossi, A., Varotto, L., Macor, F., Martina, S., Pessina, A. And Palu, C. (1989) ‘Blood pressure changes during heavy-resistance exercise’ J Hypertens Suppl, Vol. 7(6): S72-3. doi:
10.1097/00004872-198900076-00032.
Pasta, A.; Formisano, E., Calabrese, F., Plaz Torres, M.C., Bodini, G., Marabotto, E., Pisciotta, L., Giannini, E.G., Furnari, M. and Cheung, W (Ed) (2024) ‘Food Intolerances, Food
Allergies and IBS: Lights and Shadows’, Nutrients, Vol.16(2): pp265. https://doi.org/10.3390/nu16020265
Peters, D., Vadásza, I., Wujaka, L., Wygreckac, M., Olschewskid, A., Beckera, C., Herolda, S., Papp, R., Mayera, K., Rummela, S., Brandesf, R., Günthera, A., Waldeggerg, S., Eickelbergh, O., Seegera, W. and Mortya, R. (2013) ‘TGF-β
directs trafficking of the epithelial sodium channel ENaC which has implications for ion and fluid transport in acute lung injury’, Proc Natl Acad Sci U S A, Vol. 111(3): pp374–383. doi: 10.1073/pnas.1306798111
Podstawski, R., BorysBawski, K., Clark, C., Choszcz, D., Finn, K. and Gronek, P. (2019) ‘Correlations between Repeated Use of Dry Sauna for 4 x 10 Minutes, Physiological Parameters, Anthropometric Features, and Body Composition
in Young Sedentary and Overweight Men: Health Implications’, Biomed Res Int, Vol. 21(7535140): doi: 10.1155/2019/7535140. eCollection 2019.
Pscherer, S., Freude, T., Forst, T., Nussler, A., Braun, K and Ehnert, S. (2013) ‘Anti-diabetic treatment regulates pro-fibrotic TGF-β serum levels in type 2 diabetics’, Diabetol Metab
Syndr, Vol. 5(1): 48. doi: 10.1186/1758-5996-5-48
Ramakrishnan, M., Saiprasad, S. and Savaiano, D. (2024) ‘Prolonged Consumption of A2 β-Casein Milk Reduces Symptoms Compared to A1 and A2 β-Casein Milk in Lactose Maldigesters: A Two-Week Adaptation Study’, Nutrients,
Vol. 16(12): pp1963. doi: 10.3390/nu16121963
Rhee, S. (2014) ‘Lipopolysaccharide: Basic Biochemistry, Intracellular Signaling, and Physiological Impacts in the Gut ’, Intest Res, Vol.12(2): pp90-95.
Rittirsch, D., Flierl, M., Nadeau, B., Day, D., Huber-Lang, M., Grailer, J., Zetoune, F., Andjelkovic, A., Fasano, A. and Ward, P. (2013) ‘Zonulin as prehaptoglobin2 regulates lung
permeability and activates the complement system’, Am J Physiol Lung Cell Mol Physiol, Vol. 304(12): pp863–L872. doi: 10.1152/ajplung.00196.2012
Robertson, R. (2004) ‘Chronic oxidative stress as a central mechanism for glucose toxicity in pancreatic islet beta cells in diabetes’, J Biol Chem, Vol. 279(41): pp42351-42354. DOI 10.1074/jbc.R400019200
Romem, A., Lammers, K., Iacono, A., Tulapurkar, M., Dranchenberg, C., Hasday, J. and Fasano , A. (2012) ‘Zonulin - A Novel Player In Human Lung Pathophysiology’, Am J Respir Crit Care Med, Vol. 2012(185): A4266.
DOI:10.1164/ajrccm-conference.2012.185.1_MeetingAbstracts.A4266
Sant'Ana, L., Scartoni, F., Cruz, T., Ribeiro, A., Reis, N., Vieira da Silva, J., Costa Campos, Y., Araujo, G., Monteiro, S., Machado, S., Castro, A., Novaes, J. and Vianna, J. (2022) ‘Acute Effects of Different Sprint Intervals on Blood Pressure,
Heart Rate Variability, Lactate and Performance Responses in Physically Active Men’, The Open Sports Sciences Journal, Vol. 15: pp1-11. DOI:10.2174/1875399X-v15-e2102260
Sanvito, F., Nichols, A., Herrera, P., Huarte, J., Wohlwend, A., Vassalli, J. and Orci, L. (1995) ‘TGF-beta 1 overexpression in murine pancreas induces chronic pancreatitis and, together with TNF-alpha, triggers insulin-dependent
diabetes’, Biochem Biophys Res Commun, Vol. 217(3): pp1279-1286. doi: 10.1006/bbrc.1995.2906.
Sartori, C. and Matthay, M. (2002) ‘Alveolar epithelial fluid transport in acute lung injury: new insights’, Eur Respir J, Vol 20(5): pp1299–1313. DOI: 10.1183/09031936.02.00401602
Schnader, J., Juan, G., Howell, S., Fitzgerald, R. and Roussos, C. (1985) ‘Arterial CO2 partial pressure affects diaphragmatic function’, J Appl Physiol, Vol. 58(3): pp823-9. doi: 10.1152/jappl.1985.58.3.823.
Schulte, D., Küppers, V., Dartsch, N., Broermann, A., Li, H., Zarbock, A., Kamenyeva, O., Kiefer, F., Khandoga, A., Massberg, S., and Vestweber, D. (2011) ‘Stabilizing the VE cadherin-catenin complex blocks leukocyte extravasation and
vascular permeability’, EMBO J, Vol. 30(20): pp4157–4170. doi: 10.1038/emboj.2011.304.
Seiler, S. & Tønnessen, E. (2009) ‘Intervals, thresholds, and long slow distance: the role of intensity and duration in endurance training’, Sportscience, Vol. 13: pp32-53. 2009 (sportsci.org/2009/ss.htm)
Shelmet, J., Reichard, G., Skutches, C., Hoeldtke, R., Owen, O. and Boden, G. (1988) ‘Ethanol causes acute inhibition of carbohydrate, fat, and protein oxidation and insulin resistance’, J Clin Invest, Vol. 81(4): pp1137–1145. doi:
10.1172/JCI113428
Snyder, E., Beck, K., Hulsebus, M., Breen, J., Hoffman, E. & Johnson, B. (2006) ‘Short term hypoxic exposure at rest and during exercise reduces lung water in healthy humans’, J Appl Physiol, Vol. 101(6): pp1623-32. doi:
10.1152/japplphysiol.00481.2006.
Spence, A,, Naylor, L., Carter, H., Buck, C., Dembo, L., Murray, C., Watson, P., Oxborough, D., George, K. and Green, D. (2011) ‘A prospective randomised longitudinal MRI study of left ventricular adaptation to endurance and resistance
exercise training in humans’, J Physiol, Vol. 589 (Pt 22): pp5443–5452. doi: 10.1113/jphysiol.2011.217125
Sukhbir Kaur, S. and Roberts, D. (2023) ‘Why do humans need thrombospondin‑1?’, Journal of Cell Communication and Signaling, Vol. 17(3): pp485–493. https://doi.org/10.1007/s12079-023-00722-5
Tarry, D. and Powell, M. (2017) ‘Hypoxic pulmonary vasoconstriction’, BJA Education, Vol. 17(6): pp208–213, https://doi.org/10.1093/bjaed/mkw076
Taylor, J., Bonikowske, A. and Olson, T. (2021) ‘Optimizing Outcomes in Cardiac Rehabilitation: The Importance of Exercise Intensity’, Front. Cardiovasc. Med, Vol. 8:734278. doi: 10.3389/fcvm.2021.734278
Tedjasaputra, V., Bouwsema, M., and Stickland, M. (2016) ‘Effect of aerobic fitness on capillary blood volume and diffusing membrane capacity responses to exercise’, J Physiol, Vol. 594(15): pp4359-70. doi: 10.1113/JP272037.
Volynets, V., Louis, S., Pretz, D., Lang, L., Ostaff., M., Wehkamp, J., and Bischoff, S. (2017) ‘Intestinal Barrier Function and the Gut Microbiome Are Differentially Affected in Mice Fed a Western-Style Diet or Drinking Water
Supplemented with Fructose’, Comparative Study J Nutr, Vol.147(5):pp770-780. doi: 10.3945/jn.116.242859
Wahl, P., Jansen, F., Achtzehn, S., Schmitz, T., Bloch, W., Mester, J. and Werner, N. (2014) ‘Effects of High Intensity Training and High Volume Training on Endothelial Microparticles and Angiogenic Growth Factors’, Vol. 9(4): e96024.
doi:10.1371/journal.pone.0096024
Weissler, K. and Frischmeyer-Guerrerio, P. (2020) ’Genetic Evidence for the Role of Transforming Growth Factor-β in Atopic Phenotypes’, Curr Opin Immunol, Vol. 1(60): pp54–62. doi: 10.1016/j.coi.2019.05.002
Weissmann, N., Dietrich, A., Fuchs, B., Kalwa, H., Ay, M., Dumitrascu, M., Olschewski, A., Storch, U., Schnitzler, M., Ghofrani, H., Schermuly, R., Pinkenburg, O., Seeger, W., Grimminger, F. and Gudermann, T. (2006)
‘Classical transient receptor potential channel 6 (TRPC6) is essential for hypoxic pulmonary vasoconstriction and alveolar gas exchange’, PNAS, Vol. 103(50): pp19093-19098. www.pnas.orgcgi doi 10.1073pnas.0606728103
Wen, X., Zhang, B., Wu, B., Xiao, H., Li, Z., Li, R., Xu, X. and Li, T. (2022) ‘Signaling pathways in obesity: mechanisms and therapeutic interventions’, Signal Transduct Target Ther, Vol. 7(1): pp298. doi: 10.1038/s41392-022-01149-x.
Winkle MJ, Sankari A. Respiratory Muscle Strength Training. [Updated 2024 May 1]. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2024 Jan-. Available from: https://www.ncbi.nlm.nih.gov/books/NBK603753/
Yener, S., Demir, T., Akinci, B., Bayraktar, F., Kebapcilar, L., Ozcan, M., Biberoglu, S. and Yesil, S. (2006) ‘Transforming growth factor-beta 1 levels in women with prior history of gestational diabetes mellitus’, Diabetes Res Clin Pract,
Vol.76(2): pp193–198. doi:10.1016/j.diabres.2006.08.014
Zavorsky, G. (2007) ‘Evidence of pulmonary oedema triggered by exercise in healthy humans and detected with various imaging techniques’, Acta Physiol (Oxf), Vol. 189(4): pp305-17. doi: 10.1111/j.1748-1716.2006.01660.x.
Zheng, D., Zheng, D., Liao, H., Liao, H., Chen, S., Liu, X., Liu, X., Mao, C., Mao, C., Zhang, C., Zhang, C., Meng, M., Wang, Z., Wang, Y., Jiang, Q., Xue, Y., Zhou, L. and Chen, Y. (2021) ‘Elevated Levels of Circulating Biomarkers Related to
Leaky Gut Syndrome and Bacterial Translocation Are Associated With Graves’ Disease’, Front. Endocrinol, Vol. 12: https://doi.org/10.3389/fendo.2021.796212
Zouhal, H., Jacob, C., Delamarche, P., and Gratas-Delamarche, A. (2008) ‘Catecholamines and the effects of exercise, training and gender’, Sports Med, Vol. 38(5): pp401-23. doi: 10.2165/00007256-200838050-00004.
Zuttion, M., Moore, S., Chen, P., Beppu, A. and Hook, J. (2022) ‘New Insights into the Alveolar Epithelium as a Driver of Acute Respiratory Distress Syndrome’, Biomolecules, Vol. 12(9), 1273; https://doi.org/10.3390/biom12091273

Comments